Schematic
diagram for the simple electronic controller shown in the drawing above, with
an explanation of how it works.
Parts
list for the electronic stuff. (Two or three 8-pin ICs and some passive components,
available at Radio Shack. That's it!)
Introduction
I was frustrated by the limitations
of the usual Do It Yourself CO2 generating method, in which yeast growing in a bottle
of sugar water produces CO2 according to its age, temperature, and mood. This
simply doesn't allow enough control over the gas output. If you have a small
tank or soft water, uncontrolled CO2 input can cause a precipitous fall in pH when
the yeast are feeling frisky. If the yeast are old, cold, or lazy, you get
lackluster performance and a rising pH. The goal here was to build an enhanced
version of the traditional DIY CO2 system that while still cheap and fairly simple,
provides MUCH greater control over CO2 output, and that even allows for control
by pH measuring equipment should one eventually choose to do so. Having a
reservoir of CO2 in the balloon means that much less vigilance over the fermentation
process is required. There are also no expensive and hard to find high pressure
cylinders, regulators, needle valves, or solenoid valves. And there are no
trips to the welding supply shop for refills.
The Basic Setup
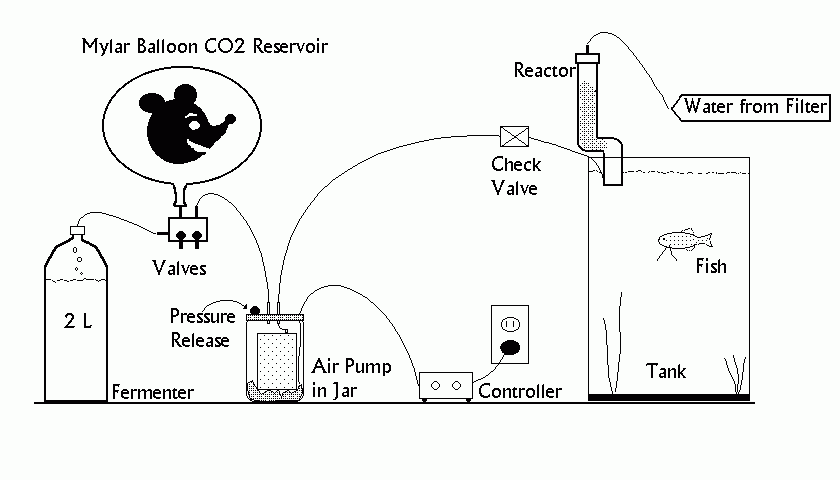
As shown in the diagram above, the
system consists of the standard two liter soda pop bottle fermenter, connected to
an air pump distribution manifold (from the fish store, marked "Valves",
above) with air line tubing. This manifold feeds both a Mylar balloon (the
reservoir), and a small aquarium air pump. The pump receives its AC power
via the electronic Controller, which is built from a couple of readily available
555 timer ICs (or one 556 dual timer) connected to a solid state relay.
Including the $8 DC adapter, this amounts to about ten dollars worth of parts, available
at any Radio Shack store, as shown in the schematic diagram. This controller provides variable control over both the length of time
the air pump runs and the interval between the pumping bursts. In my current
configuration, the pump sends half-second bursts of gas to a small gravel-filled
PVC pipe "reactor" every 40 seconds. This keeps my KH=2, 60 gallon
tank at a pH 6.6, for a calculated CO2 concentration of about about 15 parts per
million. (Unfortunately, knowing a little water chemistry like this seems to be
necessary to do CO2 supplementation properly. It's not very difficult, and
it's all well explained at the 3 aquarium sites listed below. Anyone thinking
about CO2 enrichment is strongly encouraged to do a little reading about water,
pH, buffering and the like first.)
The Balloon Reservoir
Since Mylar balloons leak Helium so
slowly, I'm assuming that CO2 molecules, being larger, will pass through even
more slowly, and that air will be slow to pass into the balloon gas reservoir from
the outside. How much this diffusion of air and CO2 through the Mylar and
attached tubing would affect the reservoir CO2 concentration over a long period
of time is unknown, but it hasn't been a problem so far. Needless to say,
a rubber balloon won't do; it's simply too porous. Mylar balloons seem
to have some sort of valve mechanism (Mylar flaps that push shut under pressure)
inside the balloon so that gas can flow in but not out. Be sure to place the tubing
into the balloon far enough so it gets past this valve -- about 6 inches or so.
If the tubing is in far enough, air blown into the balloon will be come back out
when the balloon is squeezed. Seal the tubing in place with aquarium sealant,
Shoe Goo, or whatever.
Pressure Release
It has been pointed out that if the
pressure in the fermenter/balloon/lines gets too high, the CO2 would probably be
driven through the pump and directly into the tank (i.e. you would be back to the
standard DIY CO2 setup, in which the CO2 pressure controls the dosage.) To avoid
this, some sort of pressure release mechanism is needed. In my current setup,
the tubing is just press-fit into holes in the pump enclosure. If the pressure from
the fermenter should get too high (i.e. the balloon is completely full) the excess
CO2 just leaks out around the press-fit lines. I have never actually had excess
pressure build up, except when I've squeezed the balloon to see what would happen.
This is because I use the standard 2 cups sugar, 1 tsp yeast and water mix, so the
gas production is never too far ahead of the usage -- usually the balloon is about
half full. A better solution would be to have a quarter-inch (5mm) hole in the pump
enclosure (or the top of the fermenter) and cover it with a big ball bearing or
a marble. The fermenter/balloon side of the system requires almost no pressure
at all (that's the pump's job), so loose tubing connections or a simple ball bearing
release valve will work fine to prevent too much pressure from building up.
|
|
The Air Pump and Its Enclosure
Even though my air pump has a clearly
visible intake hole and outlet port (leading me to originally believe that it was
airtight), it had to be placed in a sealed container to keep air from leaking into
the pump housing and mixing with the CO2. Apparently, the air leaks into the pump
around the rubber base. I understand that this is typical of most diaphragm pumps,
so be forewarned. An alternative to placing the pump in a sealed container
would be to tape or seal the pump bottom to keep out the air, but this would
make pump diaphragm changes difficult. Putting the pump in a sealed container
has an added advantage: it significantly reduces pump noise, especially if it is
set on a balled-up piece of cloth to provide some mechanical isolation.
I imagine the carbonic acid produced by the CO2 is going to break down the pump
diaphragm eventually, but so far (since December of 1998, so good. A pump
rebuild kit is readily available and doesn't cost much. Given the extreme
simplicity of a aquarium diaphragm air pump, I can think of no reason for it to
be harmed by being switched on and off intermittently.
|
Putting the CO2 into the Tank: The Reactor
There are lots of ways of getting the CO2 into the water; please use
whatever method appeals to you. Feeding the gas into a power head or filter
intake is probably the simplest method, and seems to work fine for lots of people.
The reactor configuration shown in the diagram above is very efficient and keeps
most of the reactor outside of the tank, so the fish and I don't have to look at
it. A trickle of the water diverted from the canister filter return line passes
down from the top of the reactor over the gravel within. The CO2 bubbles up
into the reactor through a hole in its outlet, 2 inches below the water level. This
way there is very little back pressure on the CO2 in the supply tube coming from
the pump. It should be noted that if your check valve isn't making a perfect, airtight
seal, then water will want to climb up the supply tube when the pump is off -- there's
no back pressure there to keep this from happening. For this reason, I've
chosen to feed the CO2 into the tank at the top, where the water pressure is low
and the distance to the surface is small, so the pump doesn't have to expel a lot
of water from the tube at the beginning of each pumping burst. No air stone is required
at the outlet with this configuration, since water running over gravel provides
lots of surface area. Here's a diagram of a similar
reactor that was the inspiration for mine.
By the way, if your tank has a lot
of surface agitation (from a spray bar, for example), the CO2 you're putting into
the tank won't stay in there for long. Consider returning the water to the
tank in a manner that reduces surface agitation.
Input from External pH Measuring Equipment
The electronic controller can accept an on-off control input from a pH-measuring
device with the addition of a single wire, as explained on the
schematic page. The system as shown above
is working well enough right now, though, so I don't feel any burning need to add
the additional circuitry.
Off at Night
In my setup, the AC power for the air pump comes from the timer
that controls the lights, so CO2 delivery stops at lights out. I find that
this makes the pH much more stable in the tank overnight when CO2 uptake by the
plants ceases.
Maintenance, Yeast Recipe
Once the system is assembled and the
appropriate pumping interval and duration is set, the only maintenance required
is recharging the fermenter bottle when the balloon starts to look a little flat.
(2 cups sugar, 1 teaspoon yeast, warm water.) An advantage of this system
is that since the quantity of CO2 going into the tank isn't dependent on the state
of the fermenter, you don't have to pay very close attention to how things are going
in the bottle. You'll want to have a look at the pump every now and then to
see how the diaphragm is holding up.
Alternative Implementations: Pressurization & Valve,
Acid + Carbonate
An alternative system might be to put
a one square foot piece of plywood and some bricks over the balloon to pressurize
it, and then use a valve instead of the pump. The same timer circuitry could
control the valve's "open" duration and inter-burst interval (via a transistor
instead of a Solid State Relay). This would be a very low pressure system
(i.e. << 15psi), since you'd have to place (literally) a ton of bricks on
the square-foot plywood piece to get to the usual regulator output pressure of 15
psi (15psi*144 sq.in. = 1 ton!). The low pressure would require feeding the CO2
into the tank fairly close to the surface, just as it is in the system shown.
A system like this wouldn't require any more than 12 volts DC or so (to work the
valve), so it wouldn't require doing anything with 120 volts. This may be
a more attractive way to go for people who aren't comfortable fooling with electricity
coming right out of the wall.
Another
possibility that has been suggested is to use muriatic acid and carbonate material
to generate the gas for the reservoir.
Conclusion
The idea here was to come up with something
cheaper and simpler than the "standard" tank/regulator/solenoid valve/needle
valve setup. The absence of a high pressure cylinder may make this system a little
safer. For someone who is comfortable with low-level electronic tinkering,
it may be a viable option. The whole setup was built with about $20 worth
of parts from the hardware store and Radio Shack.
My plants
are growing like mad. The fish swim happily amidst greenery and oxygen bubbles
in an aquatic jungle.

Additional information about CO2 and
planted tanks:
-
 The
Krib Everything you ever wanted to know about freshwater aquariums.
A vast, wonderful resource. The
Krib Everything you ever wanted to know about freshwater aquariums.
A vast, wonderful resource.
 Digi-Key
Electronics Home Page and Catalog. The best mail-order electronics
supplier. Digi-Key
Electronics Home Page and Catalog. The best mail-order electronics
supplier.
People who are new to electronics,
and those whose electronic skills have grown rusty, can learn a lot from Forrest
Mims' Getting Started in Electronics.
Don't let it's simple format fool you, there's a lot of good information (as well
as fun and instructive projects) there--probably enough to teach someone new to
electronics how to build a simple project like this, anyway. For the slightly
more experienced or ambitious, Horowitz and Hill's The Art of Electronics
($50) is a wonderful once-over-lightly of the world of electronics theory and practice.
Copyright (c) 1999-2017, 66pacific.com
Contact: feedback(at)66pacific.com
|
|
![]()
![]()
